Calculation of Stereoselectivity in the Boron-Mediated Aldol Reaction of Methyl Ketones
Jonathan M.
Goodman, and
Mark D Mackey
University Chemical Laboratory, Lensfield Road, Cambridge CB2 1EW, England
Abstract:
The methyl aldol procedure presented in
ECTOC
worked well on achiral ketones, but further investigations showed that it was less
successful with chiral ketones. A recent paper by Corey
(Tetrahedron Letters, 1997, 38, 33-36) encouraged us to reinterpret our
earlier calculations on these structures
(Tetrahedron Letters, 1992, 33, 7219-7222), and to introduce an additional bonding interaction.
An new procedure for calculating the stereoselectivity of boron-mediated aldol reactions
will be presented, and the results of this new approach will be evaluated.
E-mail to Jonathan Goodman


Index
Introduction
Organometallic reagents have many applications in stereoselective carbon-carbon
bond-forming reactions in organic synthesis. Only a few of these reactions, however,
may be readily modelled to analyse and predict the stereoselectivity.
One such reaction is the boron-mediated aldol reaction, which has an important place in
asymmetric organic synthesis [1]. A computational model
has been developed for this reaction, which successfully predicts the
stereoselectivity of the aldol reactions of E and Z enol borinates derived from
ethyl ketones [2], by calculating the structures and relative energies
of the competing diastereomeric transition states. A similar model has also been developed
for the addition of allyl- and crotylboranes to aldehydes [3].
Boron is particularly suited to this approach, because is cannot be more than four-valent,
and ab initio calculations on boron-containing species are relatively straightforward,
as it is in the first row of the periodic table. A similar approach could not easily be applied
to the
related reactions of allyl-tin compounds
[4], nor other allyl-metal species, because of the difficulty of performing precise molecular orbital calculations, and because of the limitations of the molecular mechanics methods which were employed.

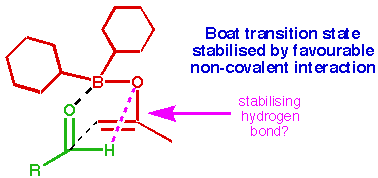
Figure 1 Reversal in stereoselectivity for methyl ketones
We have been puzzled, however,
by the behaviour of enol borinates derived from methyl ketones, which show a
reversal in stereoselectivity [5] when reacted
with 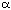 -pinene derived
chiral ligands on the boron (Figure 1).
This is not predicted by the computational model. Earlier work [6]
shows that an empirical correction (neglecting some of the transition states) greatly
improves the force field when applied to methyl ketones, but this does not account for the
behaviour of enol borinates derived from chiral ketones. In addition, the empirical correction
has no physical basis, and so is hard to interpret. In this work, we propose a new procedure for analysing the behaviour of enol borinates derived from methyl ketones, and analyse its results.
-pinene derived
chiral ligands on the boron (Figure 1).
This is not predicted by the computational model. Earlier work [6]
shows that an empirical correction (neglecting some of the transition states) greatly
improves the force field when applied to methyl ketones, but this does not account for the
behaviour of enol borinates derived from chiral ketones. In addition, the empirical correction
has no physical basis, and so is hard to interpret. In this work, we propose a new procedure for analysing the behaviour of enol borinates derived from methyl ketones, and analyse its results.





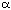 -pinene derived
chiral ligands on the boron (Figure 1).
This is not predicted by the computational model. Earlier work [6]
shows that an empirical correction (neglecting some of the transition states) greatly
improves the force field when applied to methyl ketones, but this does not account for the
behaviour of enol borinates derived from chiral ketones. In addition, the empirical correction
has no physical basis, and so is hard to interpret. In this work, we propose a new procedure for analysing the behaviour of enol borinates derived from methyl ketones, and analyse its results.
-pinene derived
chiral ligands on the boron (Figure 1).
This is not predicted by the computational model. Earlier work [6]
shows that an empirical correction (neglecting some of the transition states) greatly
improves the force field when applied to methyl ketones, but this does not account for the
behaviour of enol borinates derived from chiral ketones. In addition, the empirical correction
has no physical basis, and so is hard to interpret. In this work, we propose a new procedure for analysing the behaviour of enol borinates derived from methyl ketones, and analyse its results.